Targeted Expanded Genomic NIPT: a curated cfDNA test for serious, actionable conditions, the best screening option for increased NT

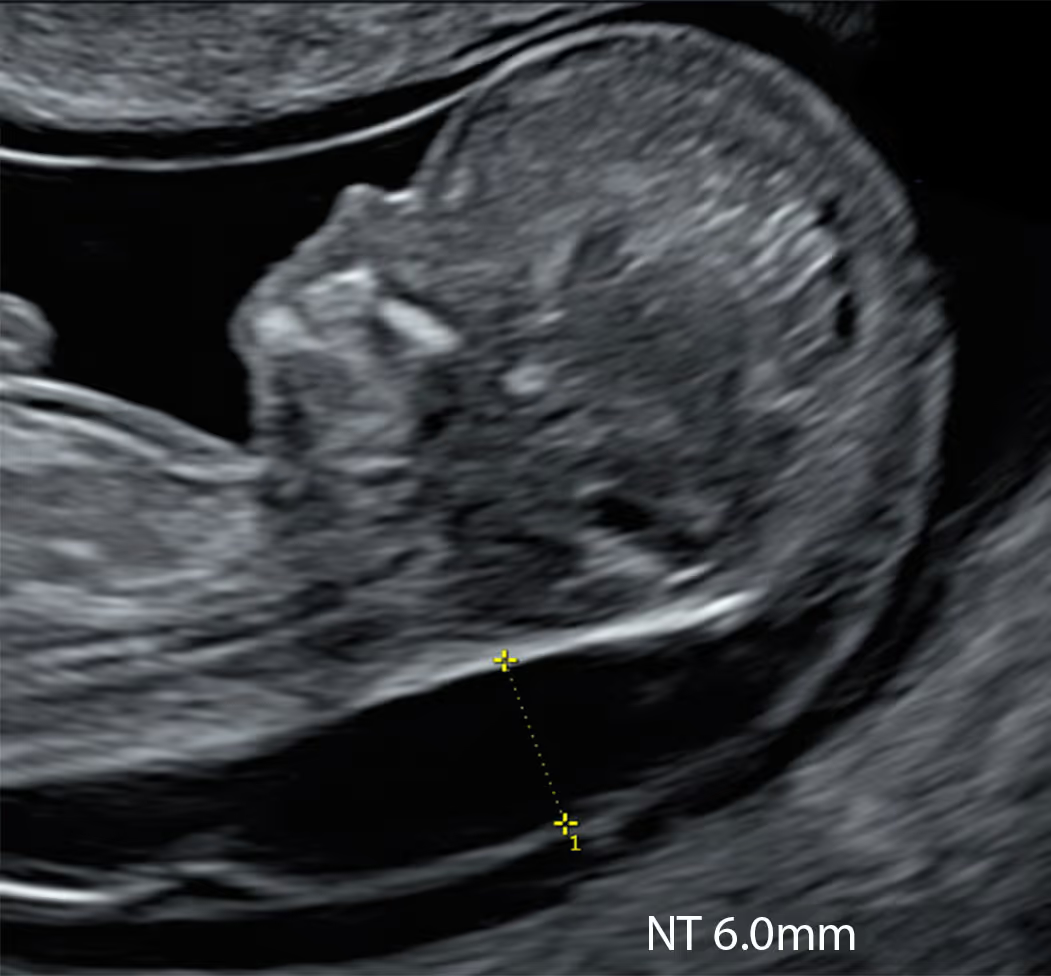
Provides high-accuracy screening for the common and uncommon aneuploidies associated with increased NT, including Down, Edwards, Patau and Turner (45X) syndromes.
Screens for 12 microdeletion syndromes; some of these can be associated with increased NT, particularly 22q11 deletion syndrome, 1p36 deletion syndrome and 18p deletion syndrome.
Most genes on this panel are strongly linked to increased NT, especially RASopathies (including Noonan syndrome), Kabuki syndrome, skeletal dysplasias and CHARGE syndrome
If KNOVA reports “low risk” for a condition/mutation it screens, it is very unlikely that the baby is affected by that condition or mutation
Here’s how KNOVA perfectly slots into the SMART NT pathway.
KNOVA offers the broadest panel of any NIPT, screening for both chromosomal and monogenic conditions. It’s supported by Fulgent’s industry-leading sequencing platform, capable of analysing over 18,000 human genes.
KNOVA uses a novel genomic approach for cfDNA analysis, which differs from all previously developed NIPTs. It is a third-generation NIPT technology called coordinative allele-aware target-enrichment sequencing (COATE-seq). This method combines aspects of previous cfDNA approaches and incorporates multidimensional analysis.
Unlike “genome-wide (GW) NIPTs”, which perform shallow analysis across all chromosomes, KNOVA uses deep next-generation sequencing targeted to about 2,000 critical loci of the human genome. This approach improves accuracy and lowers the chance of inconclusive results.
As a result, KNOVA is technologically much more advanced compared with previous NIPT brands; however, the test is less validated because it is very new (launched in 2024) and its clinical application is still limited.
While KNOVA is the most complete prenatal screening available, it isn’t suitable for every pregnancy. KNOVA cannot be used in the following situations:
KNOVA is also a screening test, not a diagnostic one. If any result shows a high risk, you may still need further testing (like CVS or amniocentesis) to confirm the result.
KNOVA Prenatal Screening (NIPT/NIPS) Full Panel (data from Fulgent Genetics):
Aneuploidies: 45X, 47XXX, 47XXY, 47XYY, Trisomy 13, Trisomy 15, Trisomy 16, Trisomy 18, Trisomy 21, and Trisomy 22
Microdeletions: 11q23q25 del, 15q11.2-q13 del, 17p11.2 del, 18p del, 18q22q23 del, 1p36 del, 22q11.2 del, 2q33 del, 4p16 del, 5p15 del, 8q23q24 del, and 9p del
Monogenic: ASXL1, BRAF, CBL, CD96, CDKL5, CHD7, COL10A1, COL11A1, COL1A1, COL1A2, COL2A1, EBP, EFNB1, ERF, FGFR1, FGFR2, FGFR3, FLNB, FREM1, GLI3, HDAC8, HNRNPK, HRAS, KAT6B, KMT2D, KRAS, LMNA, MAP2K1, MAP2K2, MECP2, NIPBL, NRAS, NSD1, NSDHL, PTPN11, RAD21, RAF1, RIT1, RUNX2, SHOC2, SKI, SLC25A24, SMC1A, SMC3, SNRPB, SOS1, SOS2, SOX9, SPECC1L, STAT3, TCF12, TRAF7, TSC1, TSC2, TWIST1, and ZIC1
KNOVA is a distinct, third-generation cfDNA platform, so there’s no true like-for-like substitute.
If unavailable, the closest options are PrenatalSafe Complete Plus (Eurofins Genoma, Italy) or a combined pathway of Panorama + Vistara (Natera, US). Both rely on more traditional technologies and, in our experience, offer narrower conditions coverage, less curated panels with higher chance of false positive/inconclusive results, longer turnaround, and (paradoxically!) often higher cost due to a less efficient workflow. (Any high-chance result still requires confirmation by CVS/amniocentesis.)
Yes, definitely. A normal NT does not rule out genetic conditions, and KNOVA still adds value:
KNOVA is a complex genomic test, and parents benefit from clear guidance. A specialist clinic should provide pre-test counselling to explain what KNOVA can and cannot reveal, an expert 10 Week Scan to confirm suitability (and to identify situations where NIPT is not helpful), and specialist genetics input to interpret any high-chance result.
If a result is high-chance, the pathway should include a detailed 12–13-week scan (with early fetal echocardiography) to refine the prenatal phenotype, followed by referral to targeted diagnostic testing CVS or amniocentesis - directed to the specific finding.
Any fetal medicine clinic offering KNOVA should work closely with Fulgent Genetics to ensure smooth logistics, accredited processing, clear reporting, and rapid support for redraws or complex results.
NT scan and NIPT at 10 weeks, then full results by week 12
1
If increased NT is found, you can have the SMART Test straight away
2
Receive genetic results in 2 weeks
3
Top-to-toe expert fetal examination
Disclaimer! These reflections are my own and arise from practice rather than committee. They do not represent the positions of the FMF, UCLH, NHS, RCOG, ISUOG, or any other organisation. As with any clinician, I may make mistakes or misjudgements. In my view, a carefully performed 10 Week Scan, used alongside appropriately selected modern NIPT and followed by targeted diagnostic testing when indicated, can provide a balanced clinical approach by supporting earlier risk assessment while avoiding unnecessary intervention or over-interpretation. Dr Fred Ushakov
KNOVA is a relatively new, advanced form of NIPT, so it isn’t yet offered everywhere. In most cases it’s arranged through a limited number of clinics that can also provide the right pre-test ultrasound checks (1st trimester anomaly scans) and appropriate counselling, which is particularly important when NT is increased.
In the UK, the clinic with the most established experience using KNOVA specifically in the setting of increased NT is London Pregnancy Clinic (LPC), where KNOVA is offered within a structured pathway that combines expert high-resolution ultrasound and genetic counselling (their SMART NT-style approach). They have performed large numbers of KNOVA tests and have a substantial team of genetic counsellors to support complex results and decision-making. They also have extensive experience supporting patients travelling from across Europe to London for this pathway, helped by London’s excellent connections to many European countries.
Outside the UK, availability varies by country and provider network, but KNOVA is offered in several regions including the USA, Canada, Australia and Japan. If KNOVA isn’t available locally, the key point is to ensure you still have a robust increased-NT pathway: expert ultrasound assessment, careful interpretation of results in context, and rapid access to targeted follow-up scans and diagnostic testing if indicated. If needed, your clinic can also consider alternative extended NIPT options where KNOVA is not accessible.
No blood test can “guarantee a healthy baby” because many important problems are structural (e.g., heart defects, brain anomalies) and are best detected by ultrasound rather than DNA screening. KNOVA does not screen for all genetic conditions, and it may not reliably detect every type of genetic change (for example, some forms of mosaicism or certain very small/complex chromosome changes). A “low chance” result does not rule out all causes of developmental or health problems. That’s why we strongly recommend combining KNOVA with a high-quality ultrasound scan.
SMART NT is our specialised pathway for pregnancies where the baby has an increased nuchal translucency (NT). It combines a high-resolution ultrasound assessment (often with a planned follow-up scan, depending on gestation and findings) with KNOVA. By bringing detailed imaging and extended genetic screening together early, SMART NT helps us clarify the likely cause of an increased NT and guide next steps. Because both ultrasound and NIPT are screening tools, any result suggesting a significant concern may still need confirmation with diagnostic testing (CVS or amniocentesis).
No combination of scans and genetic tests can guarantee a baby is free of all serious conditions, but they can provide the most informed risk assessment currently available.
No, “high chance” or "high risk" means the DNA pattern in the placenta is suggestive of that condition, but it is not proof. A key reason is that the placenta and baby can sometimes differ (called confined placental mosaicism), and occasionally results are influenced by other factors such as a vanishing twin or maternal DNA variation. The next step is usually a discussion with a fetal medicine specialist and/or genetic counsellor, and confirmation with CVS or amniocentesis if you want a definitive answer. We also consider targeted ultrasound assessment to look for supportive findings.
Yes, KNOVA NIPT can identify the baby’s biological sex with high accuracy, but this is completely optional. If you’d like to know, just let your antenatal care provider know, and if you’d rather not find out, they make sure it’s not included in your report.
Please be aware that KNOVA can also screen for sex chromosome conditions, and if your clinic includes this part of the report it may occasionally flag an increased chance of one of the following:
- Turner syndrome (45,X): the baby (typically a girl) has only one X chromosome, and this can be associated with increased NT, short stature and, in some cases, heart or kidney differences.
- Klinefelter syndrome (47,XXY): the baby (typically a boy) has an extra X chromosome, and this is often associated with taller stature and later fertility or learning/language difficulties, with many individuals only mildly affected.
- Triple X syndrome (47,XXX): the baby (a girl) has an extra X chromosome, and most are healthy, though some may have mild learning or speech difficulties and are often diagnosed later in life or not at all.
- XYY syndrome (47,XYY): the baby (a boy) has an extra Y chromosome, and most are healthy, though some may have mild learning, speech or behavioural differences.
If a sex chromosome condition is flagged, it is still a screening result, so genetic councellor would discuss what it means, review the ultrasound findings, and offer confirmatory testing (CVS or amniocentesis) if you want a definite answer.
At the moment, NHS-funded NIPT for trisomy 21, 18 and 13 is generally offered only in specific situations - most commonly if you have a higher-chance result from NHS combined screening (and meet the local eligibility criteria).
KNOVA NIPT is not routinely available on the NHS and is currently offered privately, through a limited number of authorised providers. If you’re considering KNOVA, it’s important to choose a clinic that can provide the right pre-test ultrasound assessment, clear counselling about what the test does and doesn’t cover, and a safe pathway for follow-up if the result is high chance.
Accuracy depends on the condition being screened, your background chance (age, scan findings, history), and technical factors such as the amount of placental DNA in the sample (fetal fraction).
KNOVA’s advanced proprietary technology (target enrichment, NGS, and bioinformatics) can deliver very strong performance for common trisomies and some single-gene conditions, but for rarer conditions (uncommon aneuploidies, microdeletions, and many single-gene conditions), results can be more variable, and false positives can occur.
Even with excellent laboratory methods, the positive predictive value (PPV) can be lower when a condition is rare, meaning a “high chance” result is not the same as a diagnosis. Using our SMART NT protocol, we interpret the result in conjunction with ultrasound and strongly recommend confirming with CVS or amniocentesis for certainty.
Ultrasound and NIPT answer different questions and are most powerful when combined. Ultrasound can detect many structural problems (heart, brain, abdominal wall, spine, limbs) that a DNA screening test may miss completely. It also confirms gestational age, viability and the number of babies, and can identify factors (such as a vanishing twin) that may affect how we interpret the blood test. A high-quality first-trimester scan is therefore an essential part of safe screening.
If you have already had a high-quality early scan with a genuine top-to-toe assessment of the baby, including particularly careful review of the heart, you can proceed directly to KNOVA NIPT without repeating the scan. If the scan has not been performed, was limited, or the heart could not be assessed well (for example due to fetal position or early gestation), we recommend having an expert ultrasound before the blood test so that results are interpreted in the right clinical context.
If KNOVA returns a high-chance result, we strongly recommend an expert targeted ultrasound for phenotype–genotype correlation. This means looking specifically for ultrasound features that can be associated with the flagged condition, which helps refine risk, guide counselling, and plan the most appropriate next steps (discussion regarding diagnostic testing such as CVS or amniocentesis).
KNOVA NIPT has one of the lowest redraw / no-call rates among commonly used NIPT tests. In our internal audit data, the no-call rate is under 1%.
A no-call usually means there wasn’t enough placental DNA in the sample (low fetal fraction) or the data didn’t meet quality thresholds for a reliable interpretation. It’s more common earlier in pregnancy, with higher maternal weight, and occasionally due to sample/technical factors, sometimes with no obvious reason.
Often, a repeat blood test after a short interval (when fetal fraction has increased) does produce a result. However, a no-call, particularly when driven by very low fetal fraction, can sometimes be associated with an increased chance of certain chromosome problems, especially trisomy 13, trisomy 18 and triploidy, so we discuss whether repeat testing, an expert ultrasound, and/or diagnostic testing (CVS/amniocentesis) is the best next step for you.
Occasionally, an unusual result may reflect maternal DNA rather than a fetal condition - for example, benign copy-number variants or maternal mosaicism. With single-gene panels, in a small minority of cases (around 1 in 500) the test may identify a pathogenic or likely pathogenic variant in the mother, even if she is healthy or has only mild/overlooked features. If a maternal variant is present, the baby has a 50% chance of inheriting it (depending on the gene and inheritance pattern), so healthcare provider would discuss what that means for the pregnancy and whether confirmatory testing is appropriate.
Results can also be influenced by certain medical histories - for example organ transplant, recent blood transfusion, or other rare biological factors - which is why clinicians ask relevant background questions as part of consent. If Lab think a finding may be maternal rather than fetal, your antenatal care provider will explain this carefully and outline sensible next steps, which may include review by a genetic counsellor and, where needed, targeted diagnostic testing. The aim is to provide clear, responsible guidance without causing unnecessary alarm.
In most cases, results are returned in around two calendar weeks (roughly 10–14 days) from the blood draw. KNOVA is processed at Fulgent’s laboratory in California, so the sample needs to be shipped by a specialist medical courier - this usually takes about 48 hours, but can be delayed by weekends, bank holidays, or transport disruption.
The analysis is technically complex and includes multiple quality-control steps, so it takes several working days once the sample arrives; rarely, if a run doesn’t meet strict laboratory controls and needs repeating, it can add a few extra days. Clinics are usually kept updated on progress, but because of the logistics and lab workflow, it’s not generally possible to fast-track the process.
For medical professionals who want to learn more about the SMART NT approach, access training, and join our clinical network.