Non-invasive prenatal testing (NIPT) is a maternal blood test that analyses placental cell-free DNA to screen for chromosomal and monogenic syndromes.

NIPT (non-invasive prenatal testing) is: a blood test that analyses placental (fetal) cell-free DNA (cfDNA) in maternal blood to screen for fetal chromosomal and selected single-gene (monogenic) conditions.
Modern genetic laboratories can distinguish placental (fetal) cfDNA from maternal DNA, analyse it, and use bioinformatic algorithms to screen for specific fetal conditions. NIPT is completely safe, as it uses just maternal blood sample, and highly accurate, but it is a screening test - not diagnostic.The placenta develops from the same fertilised egg as the baby and therefore largely shares the baby’s genetic make-up. Placental cells are continually shed and, as they break down, release small fragments of cfDNA into the mother’s blood.
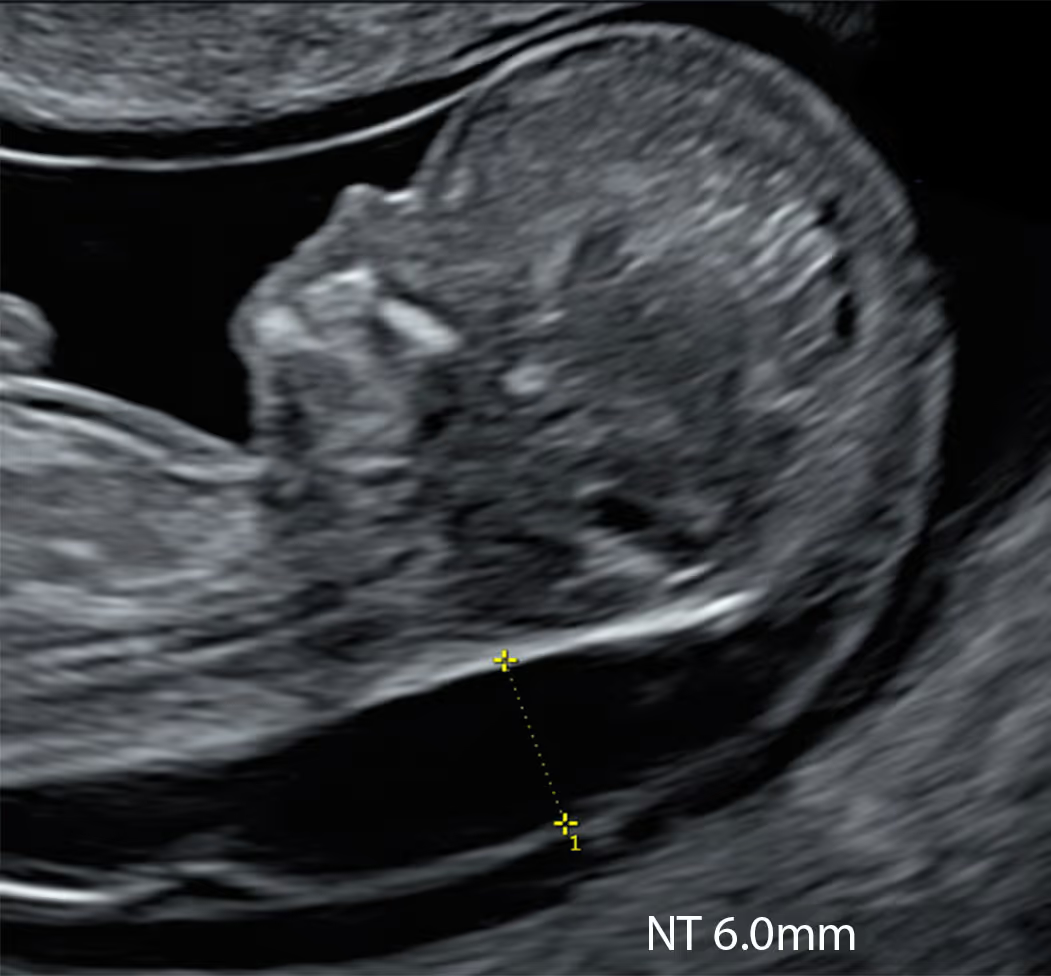
An increased nuchal translucency (NT) is not just a “chromosome screening marker.” It’s an ultrasound finding associated with a wider spectrum of outcomes: common trisomies, other chromosomal abnormalities, microdeletions, monogenic syndromes, and structural anomalies - especially congenital heart disease. Risk generally rises as the NT measurement increases.
Many conditions associated with increased NT are not detected by standard trisomy-only NIPT; however, advanced NIPT options such as KNOVA can address many of these limitations by screening for a broader range of chromosomal and genetic conditions.
Down syndrome, other aneuploidies, and sex chromosome anomalies
DiGeorge Syndrome and other copy-number variants (CNVs)
Most genetic syndromes are monogenic, such as Noonan syndrome
NIPT brand names can be confusing: different laboratories market similar tests under different trade names, and the brand alone rarely reveals the exact scope or performance without reading the fine print. When choosing NIPT, we focus on three factors: Laboratory credibility (accreditation, experience/volume, audit data, redraw policy, counselling support). Panel scope (which conditions are included: trisomies, sex chromosomes, microdeletions, and any monogenic panels; opt-in/opt-out options). Performance characteristics (sensitivity, specificity, PPV, test failure/no-call rates, fetal-fraction handling, turnaround time).
Harmony Test in the UK is a cautionary tale: a strong brand name and slick marketing eclipsed scrutiny of performance characteristics. Promoted as “the best NIPT”, it delivered only a basic panel and performed poorly, with a high no-call rate. The unreliable results affected many patients, causing anxiety and delays. The UK lab subsequently discontinued Harmony and moved to more reliable NIPT platforms. Unfortunately many UK doctors and patients still (incorrectly) refer to NIPT as “Harmony".
NIPT generally has a very high negative predictive value (NPV): i.e., when the result is reported as low risk, the probability that the fetus truly does not have the screened condition is very high. However, it remains a screening test. Positive or discordant results require confirmatory invasive diagnostic testing (chorionic villus sampling or amniocentesis), and interpretation is often complemented by phenotypic characterisation on ultrasound.


Learn More about the SMART Protocol

NIPT offers higher detection and much lower false-positive rates than CST for T21, T18 and T13. NIPT significantly reduces unnecessary invasive procedures. It’s a simple maternal blood test from ~10 weeks. Many services use a contingent approach: perform CST for all, then offer NIPT to those with a high-chance CST result (or relevant scan findings) instead of proceeding straight to CVS/amniocentesis. Any high-chance NIPT result should still be confirmed with CVS or amniocentesis.

Learn More about the SMART Protocol

NHS Genomic Medicine Service uses the term non-invasive prenatal diagnosis (NIPD). NIPD is a diagnostic test on maternal blood for specific, known monogenic conditions using validated cfDNA assays; in these cases, invasive confirmation is not required. It is considered diagnostic because it is targeted: either a bespoke, validated test for a known familial variant, or applied when the fetus shows phenotypic features strongly suggestive of a particular syndrome (e.g., FGFR3-related thanatophoric dysplasia). By contrast, basic NIPT screens for chromosomal anomalies and is not diagnostic.

Learn More about the SMART Protocol

Monogenic commercial NIPTs (e.g., KNOVA) use cfDNA methods similar to NHS NIPD and target comparable genes; however, because they are delivered as a broad screen (KNOVA currently screens 56 pathogenic genes, including FGFR3), they are classified and reported under NHS definitions as NIPT, with positives requiring traditionally invasive confirmation. In the context of increased NT, the pathway remains unclear; for example, an FGFR3-related thanatophoric dysplasia flagged on KNOVA NIPT would likely still be confirmed by targeted NHS NIPD.
An increased NT can be associated with a wide spectrum of conditions, including common trisomies, other chromosomal abnormalities, microdeletions, monogenic syndromes, and structural anomalies.
In your situation, it helps to separate “basic” NIPT from “extended” NIPT. Basic (sometimes called “core”) NIPT mainly looks for the three common chromosome conditions: Down syndrome (trisomy 21), Edwards syndrome (trisomy 18) and Patau syndrome (trisomy 13). For these three, NIPT is the most accurate screening test we have, with very high detection rates in large studies (for example, around 99% for Down syndrome) and very low false-positive rates, although results can vary a little between different test platforms and different patient groups.
Extended (expanded/advanced) NIPT tries to look beyond those three conditions. Depending on the company and the version of the test, it may also report conditions involving the sex chromosomes (such as Turner syndrome 45,X, Klinefelter syndrome 47,XXY, and others). Some versions also report rarer whole-chromosome changes (sometimes called rare autosomal trisomies) and/or “genome-wide” chromosome gains/losses, but what is included and how small a change can be detected differs a lot between providers. Some also include a limited set of specific microdeletion or microduplication syndromes (small missing or extra pieces of chromosome), and some add a panel of single-gene conditions (often called “monogenic” conditions). It is important to know that many of these expanded options have more variable performance than screening for Down/Edwards/Patau, and they do not act as a complete screen for all clinically important chromosome microdeletions/duplications.
KNOVA NIPT (by Fulgent) is an example of a broader, extended NIPT for eligible singleton pregnancies: it reports trisomy 21/18/13, may include additional trisomies (for example 15/16/22), sex chromosome conditions, selected microdeletions, and a single-gene panel described as 56 genes linked to multiple genetic syndromes. This wider scope can be relevant when the nuchal translucency (NT) is increased, because a higher NT can be associated with a broader range of genetic conditions than the three common trisomies alone, and some of the conditions on KNOVA’s single-gene panel are known to be associated with increased NT. Even so, it remains a screening test, not a diagnosis, so any “high-chance” result still needs confirmatory diagnostic testing.
Yes, this is often worth considering, and the increased NT scenario is genuinely different from routine screening. Basic (trisomy-only) NIPT is excellent for Down, Edwards and Patau syndromes, but once those are unlikely, an increased NT can still be linked to other genetic conditions, particularly certain single-gene syndromes. One of the most important groups is the RASopathies (the Noonan syndrome spectrum), which are well known to be associated with increased NT and can also be linked with cystic hygroma, fluid around the baby (hydrops/effusions) and heart findings.
Studies show that if you test specifically for RASopathies after normal chromosome results, a diagnosis is found in a small but meaningful proportion of pregnancies with isolated increased NT, and the chance is higher when the increased NT is not isolated (for example, if there are additional ultrasound abnormalities). This is exactly why a trisomy-only NIPT can feel “too narrow” for increased NT: it simply does not address this single-gene risk.
An extended NIPT that includes a monogenic (single-gene) panel, such as KNOVA, may help because it can non-invasively screen for a defined set of single-gene syndromes that are relevant to increased NT, including multiple genes linked to RASopathies and selected skeletal dysplasias. In other words, it can potentially reduce uncertainty earlier, especially for syndromes that clinicians commonly worry about when NT is increased. That said, it is still screening, not a diagnosis, and it cannot cover all single-gene disorders or all possible genetic explanations for increased NT.
It is also important to understand the limitations.
Expanded screening categories (such as rare trisomies, microdeletions and single-gene screening) have more variable performance than trisomy 21/18/13 screening, and professional guidance emphasises careful counselling and confirmatory diagnostic testing if a result is “high chance”. Single-gene NIPT is an evolving area, so results must be interpreted cautiously and always in combination with ultrasound findings.
A practical way to decide is this: if you are not ready for CVS/amnio (or you are waiting for it), an extended NIPT like KNOVA can act as a useful bridge, screening for some of the key syndromes linked to increased NT while you continue expert ultrasound follow-up. If you already plan to proceed with CVS/amnio, it may be more efficient to put resources into definitive testing on fetal DNA (such as microarray and, where appropriate, a RASopathy panel, exome or whole-genome sequencing), because that is diagnostic rather than screening.
A “no-call” result (also described as “no result”, “low fetal fraction”, or “test failure”) means the laboratory could not generate a reliable NIPT answer from the sample. No-call rates vary between different tests and different patient groups. The commonest reason is low fetal fraction (not enough placental DNA in the mother’s blood), but it can also be due to technical or quality-control issues.
A no-call matters more when NT is increased because it is not just an inconvenience or delay. Studies and clinical guidance show that test failure, especially when linked to low fetal fraction, is associated with a higher chance of chromosomal problems. In particular, persistently low fetal fraction is linked with a higher chance of trisomy 18, trisomy 13 and triploidy, which are also conditions commonly associated with increased NT. In other words, in an increased-NT pregnancy, a no-call can increase concern rather than provide reassurance.
Some no-calls happen for practical reasons, such as very early gestation, higher maternal weight, or sample/transport issues. Because time matters when you are already investigating an abnormal ultrasound marker, many clinicians will recommend prompt genetic counselling and an expert ultrasound review, and will discuss whether it is better to go straight to diagnostic testing (CVS or amniocentesis) rather than repeating the screening test and potentially losing time. A repeat blood test can still be reasonable in selected situations, but the plan should be personalised to gestation, ultrasound findings and your preferences.
Looking at the nasal bone can add a little extra information, but it does not change the main message. In Down syndrome the nasal bone is more often absent or small in the first trimester, so seeing an absent/hypoplastic nasal bone would be another finding that fits with trisomy 21, while a clearly present nasal bone would be mildly reassuring. However, once NIPT is “high chance” for trisomy 21, the result is already a very strong screening signal, and the nasal bone cannot confirm or exclude the diagnosis on its own.
The most important next step is to arrange timely genetic counselling and confirmatory diagnostic testing (CVS or amniocentesis), because only diagnostic testing can give a definite answer. Ultrasound remains important as well, not only to look at markers like the nasal bone, but to check overall anatomy and to plan follow-up (including early assessment of the heart), whatever the final genetic result turns out to be.
You should be offered an appointment with a specialist (for example a fetal medicine doctor or genetic counsellor) to explain the result in detail. The baby should be examined with an special ultrasound scan to check for any structural anomalies, especially heart defects.
You will usually be offered either NIPT (an advanced blood test) or a diagnostic test such as CVS or amniocentesis, plus follow-up ultrasounds, especially to look closely at the baby’s heart and other organs. What you choose is entirely up to you.
It is important to know that, in many countries, the term “high-chance screening result” has now replaced “high-risk screening result.”
At 10 weeks, “hydropic” usually means there is an abnormal amount of fluid around the baby - for example, generalised swelling (skin oedema), a large cystic hygroma at the back of the neck, or a significantly increased NT. This is an important finding because it can be linked to chromosomal conditions (including trisomy 21, 18, 13 and Turner syndrome), genetic syndromes (especially RASopathies such as Noonan syndrome), early heart problems, and other fetal-related causes. Very early scans can sometimes be difficult to interpret, so the first priority is to confirm exactly what is present with an expert, high-resolution scan and a clear description of the findings.
In general, diagnostic genetic tests (CVS) are not available at 10 weeks for safety reasons. Your practical options at this stage are usually to start with NIPT now (either basic NIPT or an extended option within a structured pathway such as SMART NT), and/or to plan a repeat specialist scan in 1–2 weeks when CVS may become possible. Doing NIPT now can quickly reduce uncertainty for the common trisomies, and an extended pathway such as SMART NT (high-resolution scan(s) plus KNOVA) can screen more broadly for genetic conditions relevant to increased NT/hydrops, including selected microdeletions and a panel of single-gene syndromes. This can be helpful while you are waiting to reach the gestation at which CVS can be performed.
At the same time, it is sensible to book the follow-up scan and keep the option of CVS open, because only diagnostic testing can provide a definite genetic answer.
The safest overall approach is: “do something now, and don’t lose time” - arrange an expert rescan, do NIPT promptly if you choose, and schedule the next step (repeat scan and possible CVS) in 1–2 weeks based on how the findings evolve and what the NIPT shows. If the NIPT is high chance or the ultrasound remains very concerning, you can proceed to diagnostic confirmation as soon as CVS becomes available, without unnecessary delay.
When the nuchal translucency (NT) is increased, many guidelines and expert reviews say you should be offered genetic counselling and the option of diagnostic testing (CVS or amniocentesis). The reason is simple: diagnostic tests can look for a much wider range of genetic causes than standard NIPT can. Professional guidance (including American College of Obstetricians and Gynecologists) is clear on the principle that an enlarged NT or any fetal anomaly should trigger an offer of genetic counselling, diagnostic testing, and a careful ultrasound follow-up plan.
Doctors often prioritise diagnostic testing in this situation because it can detect important chromosome changes that basic NIPT may miss. For example, chromosomal microarray (CMA) can find clinically significant small missing or extra chromosome pieces (microdeletions and microduplications, also called copy number variants - CNVs). In pregnancies with increased NT and a normal standard chromosome result, CMA can still add extra diagnoses in a meaningful number of cases. Even if NIPT comes back “low chance”, studies show there can still be clinically important chromosome findings on diagnostic testing, such as mosaicism, unusual chromosome changes, 22q11.2 deletion, and other pathogenic CNVs. The chance of these findings depends on how large the NT is and whether there are any other ultrasound concerns.
Another important group is single-gene (monogenic) syndromes that are linked to increased NT, such as Noonan syndrome and other RASopathies, Kabuki syndrome, CHARGE syndrome, and some skeletal dysplasias. These are not detected by routine chromosome tests on CVS/amnio unless the sample is specifically tested using exome sequencing (WES), whole-genome sequencing (WGS), or another targeted gene test. This is why extended screening can be attractive in the increased NT setting.
NIPT can still be a reasonable step for some families, especially if you do not want an invasive test at first. It can quickly and safely reduce uncertainty for Down syndrome, Edwards syndrome and Patau syndrome. An extended NIPT such as KNOVA can also screen for a broader range of genetic conditions linked to increased NT, including a panel of selected single-gene syndromes, which may further reduce uncertainty. However, even extended NIPT cannot cover every genetic condition and it is still a screening test, so any “high chance” result needs confirmation.
The bottom line is that with increased NT, NIPT can be part of the pathway but it usually should not be the only step. Many patients are advised to consider CVS or amniocentesis with microarray, and in selected situations exome or whole-genome testing, because these are the tests that most directly address the remaining genetic risks when NT is increased.
Positive predictive value (PPV) means: “If the test says high chance, how likely is it to be truly present?” PPV depends heavily on the chance of the condition before testing (your “starting” risk). That starting risk increases with factors such as higher maternal age and, importantly, ultrasound markers like an increased NT. As a result, when NT is increased, a “high-chance” NIPT result is more likely to be a true positive than it would be in a low-risk pregnancy.
There are two key points to understand. First, for the common trisomies (Down syndrome, Edwards syndrome and Patau syndrome), NIPT performs extremely well, so a high-chance result in an increased-NT pregnancy usually has a high PPV. Even then, NIPT is still a screening test, so the correct next step is genetic counselling and confirmatory diagnostic testing (CVS or amniocentesis) before making any major decisions.
Second, for rarer findings on expanded tests (such as certain microdeletions, rare trisomies, or small chromosome imbalances), PPV is often lower and false positives are more common. One major reason is that NIPT measures placental DNA, and sometimes the placenta has a genetic change that the baby does not (confined placental mosaicism). So, increased NT can raise the likelihood that a positive result is real, but it does not remove the need to interpret the result carefully and confirm it with diagnostic testing.
The cut-off for what is called “increased” NT varies between countries and research groups. Most researchers use the 95th centile as the threshold for increased NT, because this is the standard approach for almost every fetal measurement in medicine. NT also rises naturally as the baby grows, so what is “normal” always depends on your baby’s size.
As a simple rule of thumb, between 11 and 14 weeks an NT under about 3.5 mm is usually considered within the expected range. Measurements of 3.5 mm or more are often labelled “increased” or “high”, but in reality they represent an abnormal NT because they are above the 99th centile. At around 10 weeks’ gestation, the abnormal cut-off is lower, at about 2.5 mm, because the baby is still very small and NT is closely related to fetal size.
Your baby’s result is always interpreted in context, taking into account the exact crown–rump length, gestational age and, where relevant, the associated blood test results.
A “low chance” NIPT result for trisomy 21, 18 and 13 is reassuring for those three conditions, but it does not explain why the NT is increased. It reduces one part of the risk, but it does not remove the wider range of possible causes linked to increased NT. It’s also important to remember that, although uncommon, there is a small possibility of a false-negative NIPT result (a condition is present but the test does not detect it), for example due to low fetal fraction or true fetal mosaicism.
The main remaining (residual) risks fall into three groups. First, there can still be clinically important chromosome changes that a particular NIPT may not cover or may not detect reliably, such as certain microdeletions, microduplications, mosaicism or other atypical chromosome findings; these become more likely as the NT measurement increases. Second, there are single-gene (monogenic) syndromes, especially RASopathies (Noonan spectrum) and some skeletal dysplasias, which are well known to be associated with increased NT and are not picked up by basic chromosome screening unless specifically tested for. Third, there is the risk of structural anomalies, with congenital heart defects being one of the most important considerations in increased NT.
For this reason, the usual next steps after a low-chance NIPT with increased NT are to discuss diagnostic testing and to plan follow-up scans. Diagnostic testing by CVS or amniocentesis with chromosomal microarray is often recommended because microarray can detect clinically significant small chromosome changes that standard screening may miss. Ultrasound follow-up is also staged: a repeat expert scan (often around 14–16 weeks in many pathways) can detect a substantial proportion of structural problems earlier, and the detailed mid-trimester anomaly scan is a key reassurance point if normal. If microarray is normal but NT remains concerning, or if additional abnormalities appear, targeted testing for conditions such as RASopathies and, in selected cases, exome or whole-genome sequencing may be discussed.
This is where an extended test like KNOVA NIPT can sometimes help reduce uncertainty compared with trisomy-only NIPT. Because KNOVA may include selected microdeletions and a single-gene panel, it can potentially identify some of the genetic conditions that are more closely linked to increased NT, earlier and without an invasive procedure. However, it remains a screening test, so any “high chance” finding still needs confirmation using diagnostic testing.
It depends on what testing has been done (or is planned) on your CVS sample. If the CVS is being analysed with a comprehensive genetic approach such as chromosomal microarray plus whole-exome sequencing (WES) or whole-genome sequencing (WGS), then NIPT usually adds nothing, because you are already getting more definitive, diagnostic information directly from fetal/placental tissue. However, in many cases CVS testing is limited to rapid aneuploidy testing (for example QF-PCR) and/or microarray, which mainly assess chromosome problems and may not look for single-gene (monogenic) syndromes unless specifically requested. This matters because increased NT is strongly associated with a range of monogenic conditions, particularly RASopathies (Noonan spectrum), some skeletal dysplasias and other severe syndromes. In that scenario, KNOVA can be relevant, especially the option that focuses on its monogenic panel, because it can screen for a defined set of single-gene disorders that may not otherwise be assessed.
A practical advantage is that if KNOVA flags a specific monogenic condition, you can often use the DNA already obtained from the CVS to confirm (or refute) that exact finding, without needing another invasive procedure. The key is to review exactly what is being tested on your CVS sample and decide whether adding targeted monogenic screening (or arranging diagnostic gene testing on the CVS DNA) is the most efficient next step.
A high-chance NIPT result for trisomy 21 (Down syndrome) is a strong screening result, and the increased NT makes it even more clinically significant, but it is still not a final diagnosis. The next step is to arrange prompt counselling with an experienced clinician and plan confirmatory diagnostic testing, because only a diagnostic test can give a definite answer. Depending on gestation, this is usually CVS in the first trimester or amniocentesis from the second trimester.
At the same time, you should have an expert ultrasound scan (or a repeat high-resolution scan) to assess the baby carefully and to look for any additional features that may be associated with trisomy 21, as well as to establish a clear follow-up plan. Ultrasound cannot prove or exclude Down syndrome, but it helps with “phenotype-genotype correlation”, guides counselling, and is important for pregnancy management. Many families also find it helpful to speak to a genetic counsellor to go through what trisomy 21 can mean, the range of outcomes, and what choices are available.
While you are waiting for confirmatory results, it is sensible to plan the next steps in parallel: timing of the diagnostic procedure, what you would do with different outcomes, and what additional scans may be recommended (including a detailed heart assessment later in pregnancy). If you wish, your care team can also discuss practical support and signposting, whatever decision you make, but the priority right now is: confirm the result diagnostically and get specialist counselling quickly, without delay.
For medical professionals who want to learn more about the SMART NT approach, access training, and join our clinical network.